UPDATE: Researchers Claim New Material Can Solve Hydrogen Storage
The new metal-organic framework (MOF) has nanoscopic pores that can retain the gas.

Fuel cells bear a promise of clean transportation that is very attractive. It would just require hydrogen – the most abundant element in the universe – to combine with oxygen, generate electricity, and move cars around. But they face so many issues they are still willing to prove themselves as a viable option. Perhaps the major problem it has is how to store hydrogen more securely. Fans of the solution will be happy to hear Northwestern University researchers may have solved it.
Update: one of our readers shed some light on what the study reveals. We have updated this article to reflect that.
They had to tackle both the volumetric (size) and gravimetric (mass) deliverable capacities of hydrogen and also of methane, which can be converted to hydrogen. Their solution for that was a new metal-organic framework (MOF) with a highly porous surface that they decided to call NU-1501.
The way these researchers found to explain what it does was calling it a “programmable sponge.” In contact with NU-1501, hydrogen forms a layer over it, a process that is called adsorption.
The new MOF has almost incredible properties. One gram of it – which would have a similar volume to six M&Ms – has a surface that is equivalent to 7,310 m², or the same area of 1.3 football field.
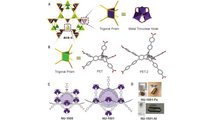
Gallery: New Metal Sponge Can Store Hydrogen At Lower Pressures
It is a pity the study's press release did not disclose exactly how much hydrogen that could represent. Anyway, one of our readers, David Martin, found part of what the original study, led by Omar K. Farha, states. Here is it:
"NU-1501-Al surpasses the gravimetric methane storage US Department of Energy target (0.5 g g−1) with an uptake of 0.66 g g−1 [262 cm3 (standard temperature and pressure, STP) cm−3] at’100 bar/270 K and a 5- to 100-bar working capacity of 0.60 g g−1 [238 cm3 (STP) cm−3] at 270 K; it also shows one of the best deliverable hydrogen capacities (14.0 weight %, 46.2 g liter−1) under a combined temperature and pressure swing (77 K/100 bar → 160 K/5 bar)."
This is how these numbers translate, according to Martin:
"For hydrogen, that means that 5 kg of hydrogen would occupy around 110 litres of space, and the MOF would weigh around 36 kg. Both metrics are way better than CF tanks. The hassle is the low temperature, which means that it is suitable for bulk storage and maybe large vehicles, especially to deliver hydrogen, not cars, unless they manage to find a way to up the temperature considerably: 77K is minus 196ºC."
The Riversimple Rasa – which has the most exciting proposition for fuel cells we have seen to date – uses 1.5 kg of the gas, which gives it 300 mi of range. So much so that Farha even considers it can be used with combustion engines, considering what he stated about the study:
“We can store tremendous amounts of hydrogen and methane within the pores of the MOFs and deliver them to the engine of the vehicle at lower pressures than needed for current fuel cell vehicles.”

We would rather see this hydrogen delivered to a fuel cell and the methane to a reformer that could feed the fuel cells with this gas. If the NU-1501 is as promising as this study suggests, we would like to see it in production as soon as possible. It could help not only the use but also the distribution of hydrogen to be much easier and safer than it is today.
It is still soon to celebrate. We will yet have to solve the hydrogen production equation: If it still comes from natural gas, for example, that’s terrible news. Anyway, the NU-1501 is encouraging for those that believe in FCEVs to be the future. Would an NU-1501 hydrogen reservoir more efficient than a battery at storing energy? How is the hydrogen released from this programmable sponge? We’ll ask the researchers and write about this fantastic solution again as soon as possible.
Source: Northwestern University via EurekAlert! and BBC
Gas storage method could help next-generation clean energy vehicles
Tremendous amounts of hydrogen and methane can be stored in nanoscopic pores
A research team led by Northwestern University has designed and synthesized new materials with ultrahigh porosity and surface area for the storage of hydrogen and methane for fuel cell-powered vehicles. These gases are attractive clean energy alternatives to carbon dioxide-producing fossil fuels.
The designer materials, a type of a metal-organic framework (MOF), can store significantly more hydrogen and methane than conventional adsorbent materials at much safer pressures and at much lower costs.
“We’ve developed a better onboard storage method for hydrogen and methane gas for next-generation clean energy vehicles,” said Omar K. Farha, who led the research. “To do this, we used chemical principles to design porous materials with precise atomic arrangement, thereby achieving ultrahigh porosity.
Adsorbents are porous solids which bind liquid or gaseous molecules to their surface. Thanks to its nanoscopic pores, a one-gram sample of the Northwestern material (with a volume of six M&Ms) has a surface area that would cover 1.3 football fields.
The new materials also could be a breakthrough for the gas storage industry at large, Farha said, because many industries and applications require the use of compressed gases such as oxygen, hydrogen, methane and others.
Farha is an associate professor of chemistry in the Weinberg College of Arts and Sciences. He also is a member of Northwestern’s International Institute for Nanotechnology.
The study, combining experiment and molecular simulation, will be published on April 17 by the journal Science.
Farha is the lead and corresponding author. Zhijie Chen, a postdoctoral fellow in Farha’s group, is co-first author. Penghao Li, a postdoctoral fellow in the lab of Sir Fraser Stoddart, Board of Trustees Professor of Chemistry at Northwestern, also is a co-first author. Stoddart is an author on the paper.
The ultraporous MOFs, named NU-1501, are built from organic molecules and metal ions or clusters which self-assemble to form multidimensional, highly crystalline, porous frameworks. To picture the structure of a MOF, Farha said, envision a set of Tinkertoys in which the metal ions or clusters are the circular or square nodes and the organic molecules are the rods holding the nodes together.
Hydrogen- and methane-powered vehicles currently require high-pressure compression to operate. The pressure of a hydrogen tank is 300 times greater than the pressure in car tires. Because of hydrogen’s low density, it is expensive to accomplish this pressure, and it also can be unsafe because the gas is highly flammable.
Developing new adsorbent materials that can store hydrogen and methane gas onboard vehicles at much lower pressures can help scientists and engineers reach U.S. Department of Energy targets for developing the next generation of clean energy automobiles.
To meet these goals, both the size and weight of the onboard fuel tank need to be optimized. The highly porous materials in this study balance both the volumetric (size) and gravimetric (mass) deliverable capacities of hydrogen and methane, bringing researchers one step closer to attaining these targets.
“We can store tremendous amounts of hydrogen and methane within the pores of the MOFs and deliver them to the engine of the vehicle at lower pressures than needed for current fuel cell vehicles,” Farha said.
The Northwestern researchers conceived the idea of their MOFs and, in collaboration with computational modelers at the Colorado School of Mines, confirmed that this class of materials is very intriguing. Farha and his team then designed, synthesized and characterized the materials. They also collaborated with scientists at the National Institute for Standards and Technology (NIST) to conduct high-pressure gas sorption experiments.
The research was supported by the U.S. Department of Energy’s Office of Energy Efficiency and Renewable Energy (award no. DE‐EE0008816).
The title of the paper is “Balancing volumetric and gravimetric uptake in highly porous materials for clean energy.”
RECOMMENDED FOR YOU

Rivian's New AI Voice Assistant Is Rolling Out. Here's What It Can Do

Android Auto’s Massive Update Brings Edge-To-Edge Maps To Every Car Screen

Rivian May Build Its Own Lidar Sensor For The R2 In The U.S.

Kia Is Ready To Build An Electric Successor To The Stinger. One Thing Is Holding It Back.

Inside Ford's $1 Billion Bet On Detroit’s Future: Newlab at Michigan Central

This Radical Beach Cruiser Is A Refreshing Cure For Boring EV Designs

The Base 2027 Mercedes C-Class EV Has Three Screens, But One Is Hiding A Secret