Membraneless Fuel Cell May Allow EVs To Use Ethanol To Recharge
Taking reformers out of the equation will allow for an easier EV adoption in the future.
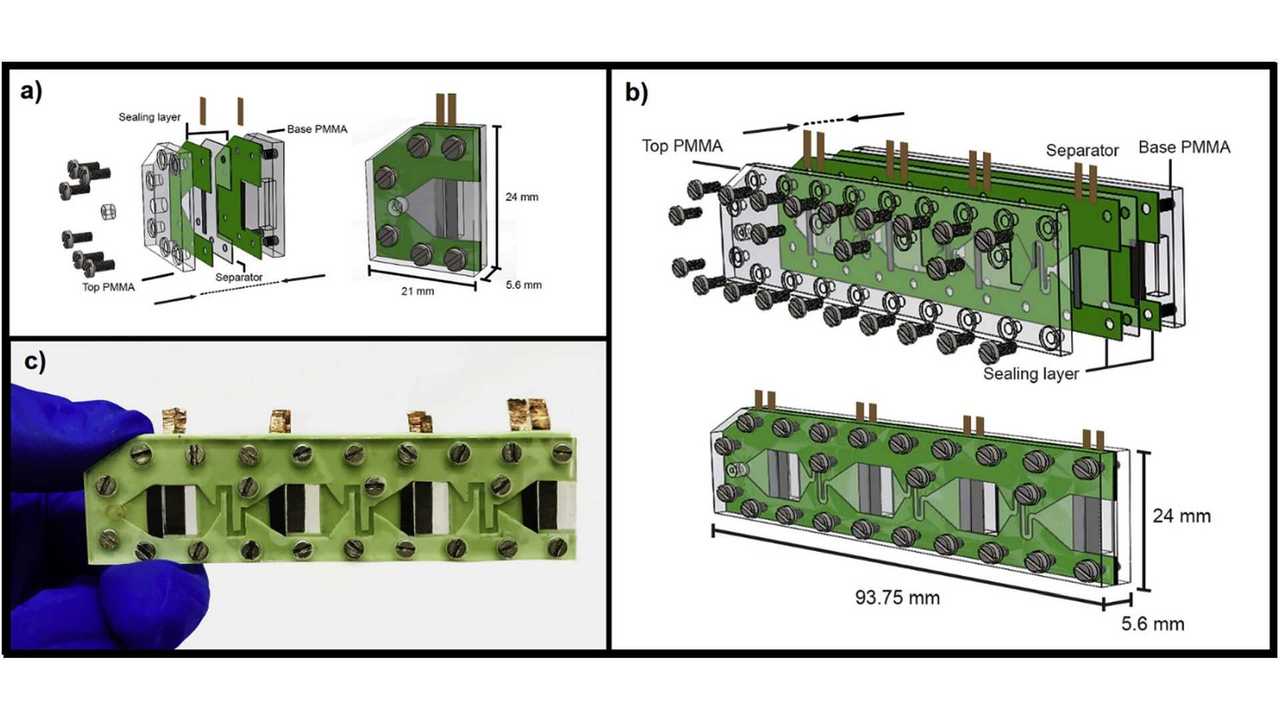
For fuel cells to make sense, you either take a completely new approach towards them – such as Riversimple did – or accept that its best application is with transportation means that are difficult to decarbonize (trucks, trains, ships, and airplanes). Large hydrogen tanks make them difficult to use in passenger cars, but scientists at INRS may have found a way to facilitate their adoption in cars: eliminating the membrane they have.
If you ask how a membraneless fuel cell could do the trick – apart from being much more affordable than regular ones – the explanation is not straightforward. For you to get it, you need to understand how fuel cells work.
The regular ones use a membrane and are called PEMFC (Proton Exchange Membrane Fuel Cells). These costly membranes, usually made of Nafion, control the reaction between oxygen and hydrogen that forms water and generates electricity. So you always need to have pure hydrogen fed to the PEMFC. You get it either from expensive high-pressure tanks or through reformers, machines that extract hydrogen from fuels.
We have already told you that the Gumpert Nathalie uses fuel cells and methanol. Nissan is testing in Brazil a fuel cell vehicle that uses ethanol to extract the hydrogen it needs. The issue with reformers is that they add complexity, cost and mass to the vehicle. That is not a problem if the fuel cell can deal with methanol or ethanol directly.

Using the oxygen already present in the air, the membraneless fuel cell manages to turn the oxidation the fuel suffers naturally into energy. The trick was using selective electrodes that do not react with the methanol or ethanol, only with oxygen.
So far, the membraneless fuel cell developed by INRS managed to power a LED for four hours using just 234 microliters (0.000234 l) of methanol. Mohamed Mohamedi, one of the study's lead authors, told InsideEVs about potential automotive applications for the new fuel cell.
“In our study, we used methanol as a benchmark molecule because we understand the electrochemical processes involved. However, parallel intensive research is being conducted in my lab towards developing catalysts that could oxidize completely ethanol (produced from biomass) to water and CO2. If we succeed in discovering these catalysts, then we may consider automotive applications of our membraneless fuel cells.”

People that see no point in FCEVs will continue to dismiss them no matter what. For the ones that know they can help decarbonize transportation, this is big news. They also understand that renewable fuels do not increase the amount of carbon in the atmosphere and, consequently, do not worsen climate change.
That said, emerging countries that cannot afford massive infrastructure investments for chargers could use the fuel pumps they already have to distribute renewable fuel to membraneless fuel cell vehicles, for example. As long as it is produced from biomass – the so-called second-generation ethanol – this could be an environmentally friendly push for EV adoption.
The ones that blindly defend BEVs at all costs probably oversee that the mining required to build all necessary batteries is an environmental concern unless we develop more sustainable batteries, such as the single carbon electrode developed by PJPEye. They also probably believe building an expensive charging infrastructure to support BEVs will be something all countries will manage to afford. Unfortunately, that’s not the case.
For electrification to be widespread, it must be affordable for everyone. Otherwise, developed countries will be proud of how green their fleet is while nearby nations burn tons of diesel in 30-year-old trucks and buses that first-world countries do not want anymore.
A successful membraneless fuel cell could avoid that. These EVs would be cheaper and would not need to be recharged, just refilled, and no additional carbon would be added to the atmosphere. If the goal is cleaning transportation means, this is something to celebrate as much as any other solution. We hope Mohamedi and his team progress swiftly with their research.
Images appear courtesy of Elsevier, Copyright Elsevier (2020).
Source: INRS via ScienceDirect
RECOMMENDED FOR YOU

Volvo Gets A Pass To Keep Importing Connected Cars To The U.S.

General Motors Is Bringing Sodium-Ion Battery Production To America

The Volvo EX60 Can 'See' The World And Chat With You About It

GM Is Making EV Charging A Lot Simpler With Energy Pass, NACS On All 2027 Models

Rivian's New AI Voice Assistant Is Rolling Out. Here's What It Can Do

Mexican EV Startup Unveils $8,600 State-Backed EV With 6 Seats

Rivian May Build Its Own Lidar Sensor For The R2 In The U.S.
